
During and after translation, the precursor protein undergoes an ordered series of proteolytic cleavages by viral and host proteases that generate the individual viral proteins. The precursor consists of N-terminal structural and C-terminal nonstructural components. The HCV genome, a single plus-stranded RNA molecule of 9,600 nucleotides, encodes a single precursor polyprotein of about 3,000 amino acids (for reviews, see references 6 and 58). Although these subgenomic replicons were recently extended to the full-length HCV genome encoding all viral proteins, no viral particle formation was observed ( 52). Important progress was made with the development of autonomously replicating HCV RNAs which produce nonstructural viral proteins upon transfection into hepatoma cells ( 7, 27). The lack of an efficient infectious cell culture model has prevented a comprehensive understanding of the viral life cycle. Analyses of viral kinetics showed that HCV particles are continuously produced and cleared during chronic infection ( 46, 54). Viral infection persists in ∼80% of infected individuals, causing chronic hepatitis, liver cirrhosis, and hepatocellular carcinoma ( 1, 13). Hepatitis C virus (HCV), the major causative agent of non-A, non-B hepatitis, is estimated to infect 3% of the world's population ( 11). The location of the core protein in the outer mitochondrial membrane suggests that it could modulate apoptosis or lipid transfer, both of which are associated with this subcellular compartment, during HCV infection. A stretch of 10 amino acids within the hydrophobic C terminus of the processed core protein conferred mitochondrial localization when it was fused to green fluorescent protein. Using immunoelectron microscopy and in vitro mitochondrial import assays, we showed that the core protein is located on the mitochondrial outer membrane. The core protein also fractionated with mitochondrion-associated membranes, a site of physical contact between the ER and mitochondria. Subcellular fractionation confirmed this observation and showed that the core protein associates with purified mitochondrial fractions devoid of ER contaminants. 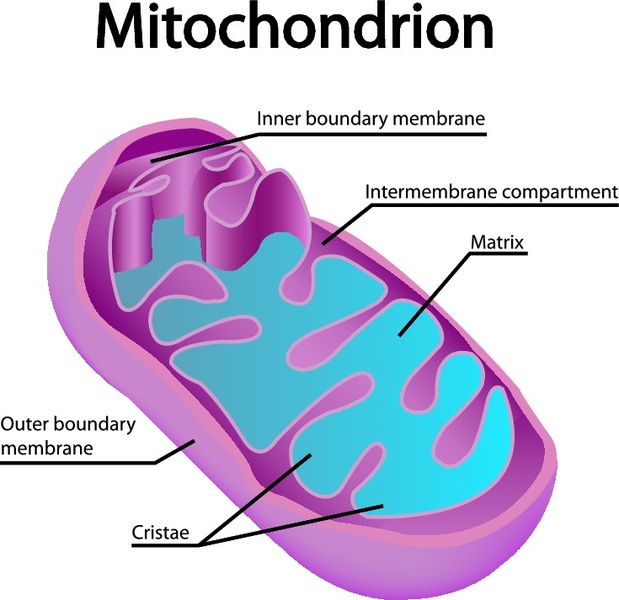
Using confocal microscopy, we observed that a fraction of the mature core protein colocalized with mitochondrial markers in core-expressing HeLa cells and in Huh-7 cells containing the full-length HCV replicon. Processing at position 179 by a recently identified intramembrane signal peptide peptidase leads to the generation and potential cytosolic release of a 179-amino-acid matured form of the core protein. 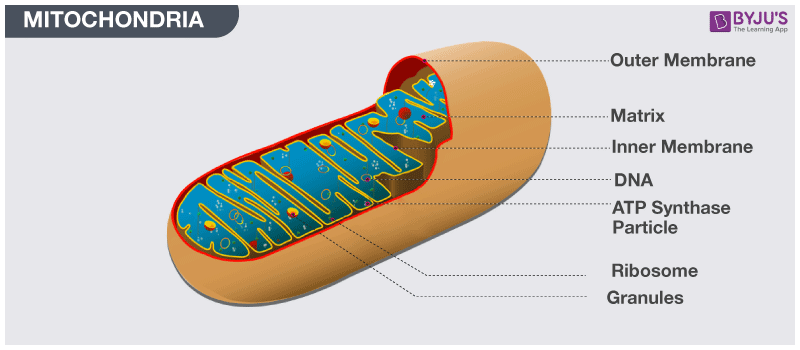
The hepatitis C virus (HCV) core protein represents the first 191 amino acids of the viral precursor polyprotein and is cotranslationally inserted into the membrane of the endoplasmic reticulum (ER).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
